 
There are five different electron-domain geometries: Linear (two electron domains), trigonal planar (three domains) tetrahedral (four domains) trigonal bipyramidal (five domains) octahedral (six domains). How many different electron domain geometries are consistent with the molecular geometry shown? In the first instance, all the regions are bonding regions and the shape of the molecule is the same as the geometry around the central atom, trigonal planar. There are two possible shapes of molecules whose geometry is trigonal planar. How many molecular geometries are there for the trigonal planar electron geometry? There are two molecular geometries that can come out of three electron domains, trigonal planar (no lone pairs) and bent with ≈120° bond angle (one lone pair). 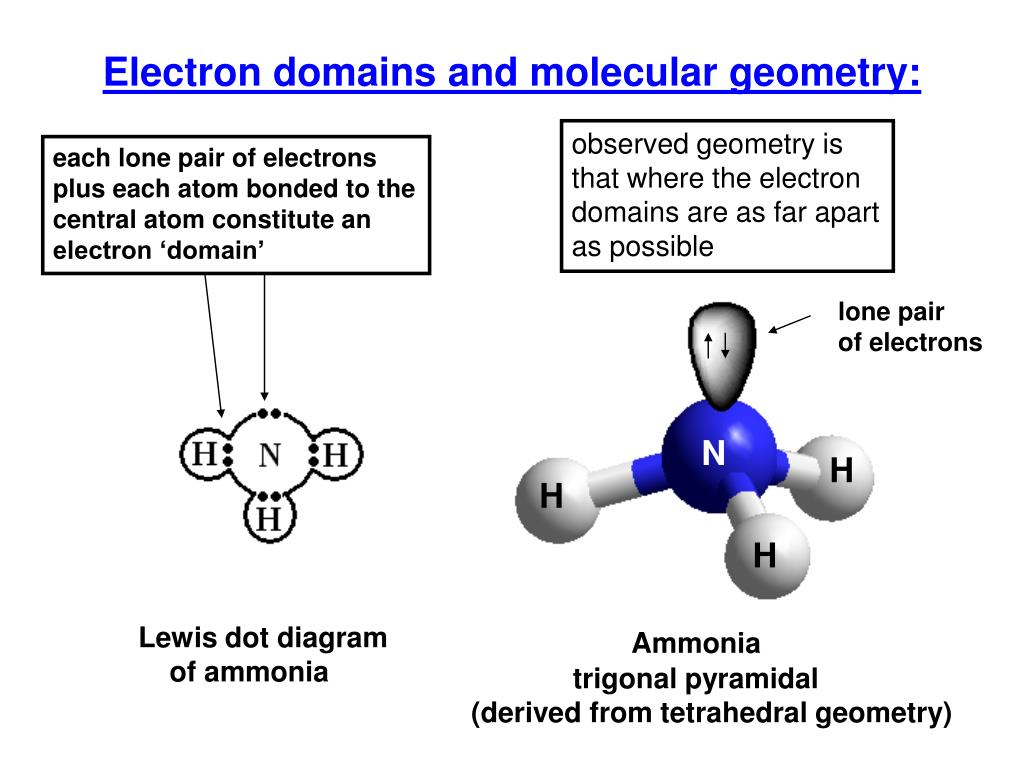
How many molecular geometries are there for the trigonal planar? The arrangement of electron pairs around a central atom is known as electron geometry. What is electron geometry vs molecular geometry?ĭifference Between Electron Geometry and Molecular Geometry The arrangement of atoms in a molecule, usually relative to a single central atom, is known as molecular geometry. One important point to note about molecular shape is that all diatomic (compounds with two atoms) compounds are linear. The five ideal shapes are: linear, trigonal planar, tetrahedral, trigonal bipyramidal and octahedral. What are the 5 major shapes of covalent molecules? The shape of the orbitals is tetrahedral. The molecule that is made up of 4 equally spaced sp3 hybrid orbitals forming bond angles of approximately 109.5o. Some of the most common shapes that can be taken are linear, trigonal planar, tetrahedral, pyramidal, and angular (or bent). What are the most common molecular geometries explain with examples? Divide the total of these by 2 to find the total.Add or subtract electrons for charge (see Top Tip).Add one electron for each bonding atom.How do you find the geometry of a compound? The 6 basic molecular shapes are linear, trigonal planar, angular (bent), tetrahedral, trigonal pyramidal, and trigonal bipyramidal. What are the 6 types of geometry applicable to organic chemistry?įor organic molecules, we will observe the same types of geometry – linear, trigonal planar, tetrahedral, trigonal pyramid, and bent. Euclidean, or classical, geometry is the most commonly known geometry, and is the geometry taught most often in schools, especially at the lower levels. Therefore, 4 different molecular geometries are possible for a compound with a central atom that has five regions of electron density. How many molecular geometries are possible for a compound? Understanding the molecular structure of a compound can help determine the polarity, reactivity, phase of matter, color, magnetism, as well as the biological activity. Molecular geometry, also known as the molecular structure, is the three-dimensional structure or arrangement of atoms in a molecule. What are the 5 molecular geometries? The 5 molecular geometries are linear, trigonal planar, tetrahedral, trigonal bipyramidal and octahedral. 24 What does VSEPR stand for? How many types of geometry are there in chemistry? 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
